S0728670
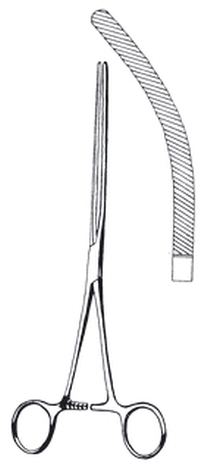
Forceps,intest,clamp,Doyen,230mm,cvd
Forceps, intestinal clamp, Doyen, 230 mm, curved
Indicative Price 4.14 USD
General Description:
Forceps, intestinal clamp, Doyen, 230 mm, curved
Intended use:
For transverse occlusion of a section of intestine while suturing, resection or ananastomosis.
Technical Specifications:
Pressure force instrument: intestinal clamp, springy
See Martin Catalogue no. 24 Ref: 30 275 23.
Non-crushing clamp, curved.
Long jaws and shorter shanks.
Smooth, springy jaws with atraumatic longitudinal serrations.
Occlusion: The form and mechanical action must permit precise closing.
Adhesion: Provided by the surface of the jaws.
Length: 23 cm.
Material: Martensitic steel (quenched, magnetic steel).
Martensitic steel composition: 0.15-0.20% carbon, 12-13% chromium.
Reusable.
Environmental conditions:
Storage and transport conditions: 1°C to 30°C.
Humidity: 30% RH to 70% RH.
Quality Management system:
ISO 13485:2016: Medical devices - Quality management systems
Classification:
EU Medical Devices Directive 93/42/ECC, Class I
Compliance to safety & product performance standards:
ISO 7153-1:2016: Surgical instruments - Materials - Part 1: Metals.
ISO 7151:1988: Surgical instruments - Non-cutting, articulated instruments - General requirements and test methods.
ISO 13402:1995: Surgical and dental hand instruments - Determination of resistance against autoclaving, corrosion and thermal exposure.
ISO 17664:2017: Processing of health care products - Information to be provided by the medical device manufacturer for the processing of medical devices.
ISO 14971:2007 Medical devices - Application of risk management to medical devices.
Nomenclature:
GMDN code: Surgical soft-tissue manipulation forceps, scissors-like, reusable (62468)
UMDNS code: Forceps, Intestinal (11785)
Safety process:
This item must be cleaned, disinfected after each use, and sterilised in a steam steriliser.
Packaging and labelling:
Primary packaging: Unit of use.
One (1) forceps in a plastic bag.
Secondary packaging: Protected unit.
Ten (10) forceps in a box.
Component of a kit:
S9910000 - Surg. inst., abdominal /SET.
Related Products