S0767500
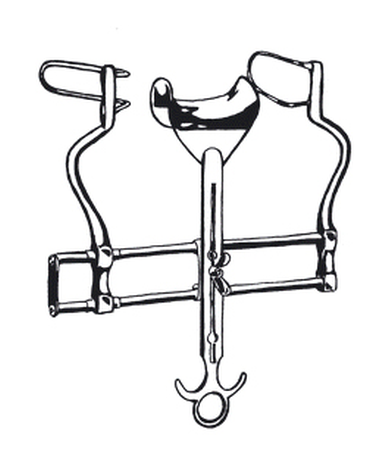
Retractor,abdo,Balfour,3 blades
Retractor, abdominal, Balfour, 3 blades.
Indicative Price 33.12 USD
General Description:
Retractor, abdominal, Balfour, 3 blades.
Intended use:
Surgical instrument to separate fatty tissue, muscles or viscera after the abdominal incision to expose the operative field for examination or intervention.
Technical Specifications:
Static function instrument: abdominal self-retaining retractor.
See Martin Catalogue no. 24 Ref: 15 852 00.
Locking mechanism and compass (self-holding).
The compass (frame) is a support with hinged arms.
2 fenestrated end blades: approx. 35 x 70 mm.
1 center blade, adjustable in height: approx. 45 x 80 mm.
Maximum wound opening: approx. 180 mm.
Material: Martensitic steel (quenched, magnetic steel).
Martensitic steel composition: 0.15-0.50% carbon, 12-15% chromium, 0.45-0.90% molybdenum.
Reusable.
Environmental conditions:
Storage and transport conditions: 5°C to 40°C.
Humidity: 40% RH to 70% RH.
Quality Management System:
ISO 13485:2016: Medical devices - Quality management systems
Classification:
EU Medical Devices Directive 93/42/ECC, Class I
Compliance to safety & product performance standards:
ISO 7153-1:2016: Surgical instruments - Materials - Part 1: Metals.
ISO 7151:1988: Surgical instruments - Non-cutting, articulated instruments - General requirements and test methods.
ISO 17664:2017: Processing of health care products - Information to be provided by the medical device manufacturer for the processing of medical devices.
ISO 14971:2007 Medical devices - Application of risk management to medical devices.
Nomenclature:
GMDN code: Self-retaining surgical retractor, reusable (45182)
UMDNS code: Retractors, Self-Retaining Operating (13390)
Safety process:
This item must be cleaned, disinfected after each use, and sterilised in a steam steriliser.
Packaging and labelling:
Primary packaging: Unit of use
One (1) retractor in a plastic bag.
Component of a kit:
S9910000 - Surg. inst., abdominal /SET.
Related Products