S0001000
Vacuum extractor,Bird(ant post),elec,set
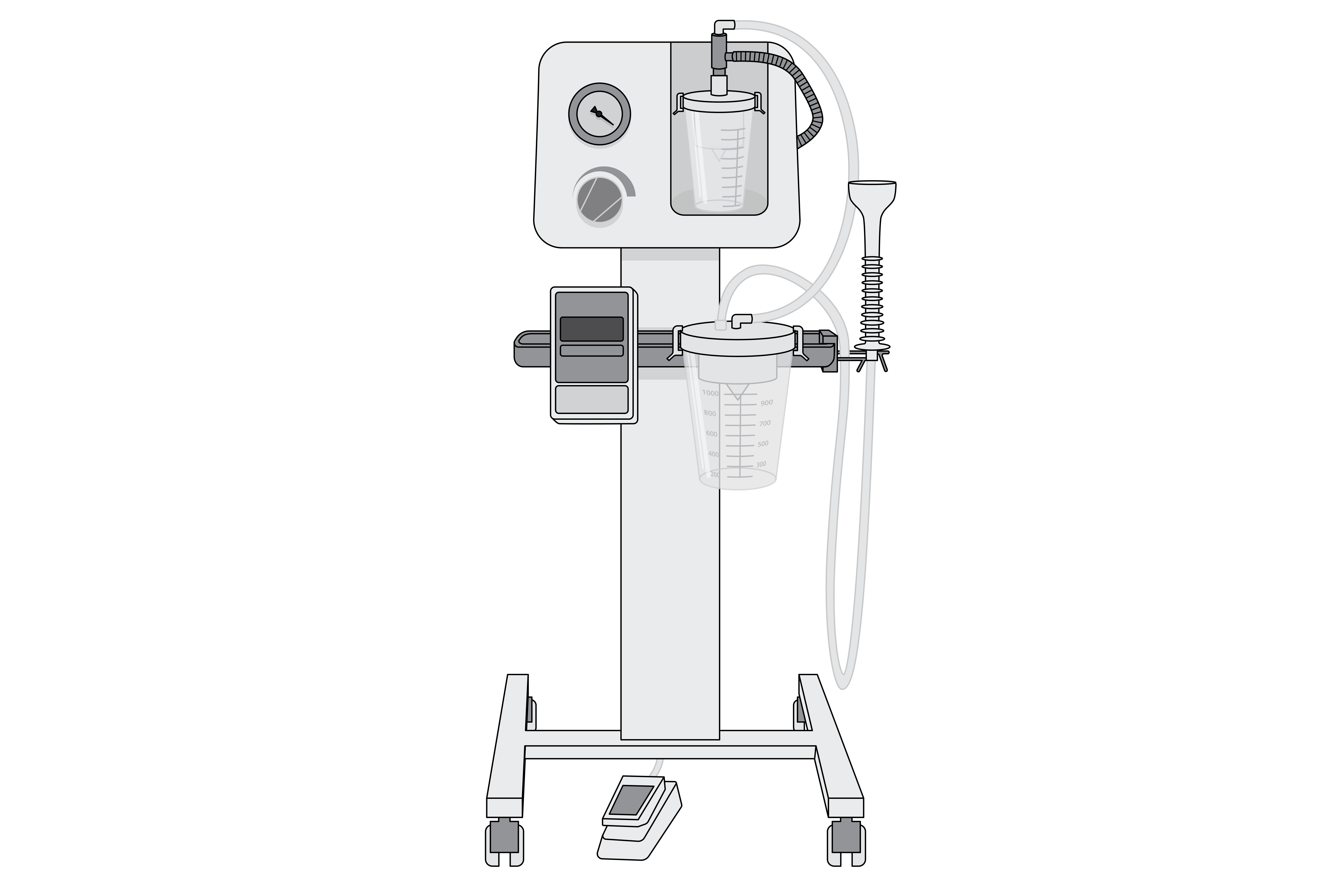
Vacuum extractor set Bird (with anterior and posterior cups) and an electrically-powered suction pump.
Indicative Price 3,768.75 USD
GENERAL DESCRIPTION
Vacuum extractor, Bird (with anterior and posterior cups), electric, complete set,
INTENDED USE
A full set to facilitate vaginal or caesarean section delivery by traction utilizing vacuum. Includes suction caps intended to be applied to the scalp of a foetus, a handgrip for the clinician to apply traction, and an electrically-powered suction pump. It is typically used for dystocia, uterine inertia, maternal exhaustion, or maternal/foetal distress. This is a reusable device.
TECHNICAL SPECIFICATIONS
Vacuum extractor system to assist vaginal deliveries in a delivery room setting.
Electrically powered vacuum/suction pump creates and maintains required vacuum.
Vacuum range, adjustable: from 0 to approx. -90 kPa/ -675 mmHg.
Gauge dial, minimal graduation: 5kPa/37.5 mmHg.
Accuracy vacuum gauge: approx. ±2.5 %.
Servo system monitors vacuum and when necessary, initiates suction to compensate for loss of vacuum.
Suction flow maximum: approx. 30L/min.
Front panel informs actual vacuum created, with dial gauge in kPa and mmHg.
With vacuum control button and on/off-switch.
With foot pedal with operate/stop switch.
Plastic (PSU, autoclavable) vacuum collection bottle, with filter keeps circuitry clean.
Capacity of vacuum collection bottle 1 Litre.
Device has sturdy shock resistant and electrically isolated housing, easy to move and easy clean.
All parts of the vacuum extractor can be disassembled.
Relevant parts can be autoclaved at 121°C.
Requires routine maintenance, manageable at the level of the delivery rooms.
Power requirements: 100 - 240V / 50Hz
SYSTEM IS SUPPLIED AS A COMPLETE SET CONSITSTING OF:
2 x Bird type anterior suction/extraction cup (1 x 50mm and 1 x 60mm diameter) rounded edges.
1 x Bird type posterior suction/extraction cup (1 x 50 mm diameter) rounded edges.
2 x Soft vacuum extractor cups "silc-cup": diameter 50mm and 60mm.
2 x Extraction handle, stainless steel.
1 x Reusable, 1 Litre vacuum collection bottle + lid, PSU, with clamp holder.
1 x Foot pedal with operate/stop switch.
1 x 5m Power cable, with wall plug.
TROLLEY
Sturdy and stable construction on 4 antistatic swivel castors, all with brakes.
Diameter castors: approx. 75mm
Carrying capacity: approx. 15kg.
Back cable holder to facilitate transport and positioning.
Trolley with standard rail for the attachment of vacuum collection bottle, accessories, and foot vacuum on/off switch.
Material: PS coated metal.
SUPPLIED WITH
Instructions for assembly, use and maintenance in English, French and Spanish
1 x Spare vacuum collection bottle.
1 x Spare tube, 3m.
20 x Polypropylene bottom plate, for stainless steel suction/extraction cup, 10 for each size, 50 and 60mm.
1 x List of accessories and parts.
ESTIMATED DELIVERY LEAD TIME
4 – 6 Weeks
ESTIMATED LIFE SPAN
Seven years.
WARRANTY
Five years for the basic pump.
ENVIRONMENTAL CONDITIONS
Storage conditions: -20 - 50°C / 20 to 95% RH.
Operating conditions: 5 - 40°C / 30 to 75% RH.
Atmospheric pressure: 70 to 106 kPa.
WEIGHT AND VOLUME (Packaged)
Weight: 16 Kg.
Volume: 236 dm³.
INSTALLATION / COMMISSIONING REQUIREMENTS
Some simple assembly will be required such as placing the main suction unit on top of the trolley and installing the suctions bottle and connecting cables and tubing. This does not necessarily have to be carried out by a qualified biomedical engineer.
TRAINING REQUIREMENTS
Vacuum extraction should only be performed or supervised by trained and experience physicians. The manufacturer does not provide clinical training on vacuum extraction. The instructions for use included with the devices provides the necessary details for physicians trained on carrying out vacuum extractions not familiar with this specific device.
MAINTENANCE/USER REQUIREMENTS
Other than cleaning in accordance with manufactures instructions there are not specific maintenance requirements.
ALTERNATIVE PRODUCTS
S0791500, Vacuum extractor,Bird,manual,comple.set
S0791501, Vacuum extractor,Bird(ant&post),manu,set
QUALITY MANAGEMENT SYSTEM
- Manufacturer is certified for ISO 13485 Medical devices - Quality management systems - Requirements for regulatory purposes.
MARKET CLEARANCE AND DEVICE CLASSIFICATION
CE certified under the EU MDR 2017/745 as Class IIa device.
SAFETY AND PRODUCT STANDARDS
- IEC 60601-1:2005+AMD1:2012+AMD2:2020 CSV Medical electrical equipment - Part 1: General requirements for basic safety and essential performance. Attached test reports or certificates confirming compliance, indicate the file name in free text cell.
- IEC 60601-1-2:2014+AMD1:2020 CSV Medical electrical equipment - Part 1-2: General requirements for basic safety and essential performance - Collateral standard: Electromagnetic compatibility - Requirements and tests . Attached test reports or certificates confirming compliance, indicate the file name in free text cell.
- ISO 10079-1:2022 Medical suction equipment -- Part 1: Electrically powered suction equipment. Attached test reports or certificates confirming compliance, indicate the file name in free text cell.
- ISO 10993-1:2018 Biological evaluation of medical devices — Part 1: Evaluation and testing within a risk management process.
- ISO 10993-5:2009 Biological evaluation of medical devices. Tests for in vitro cytotoxicity.
- ISO 10993-18:2020 Biological evaluation of medical devices. Chemical characterization of materials.
NOMENCLATURE
GMDN Code(s): 32596, 63649, 36336, 40596, 63642, 44943
Related Products