S0481057
Thermometer,clinical,non-cntct,incl bat
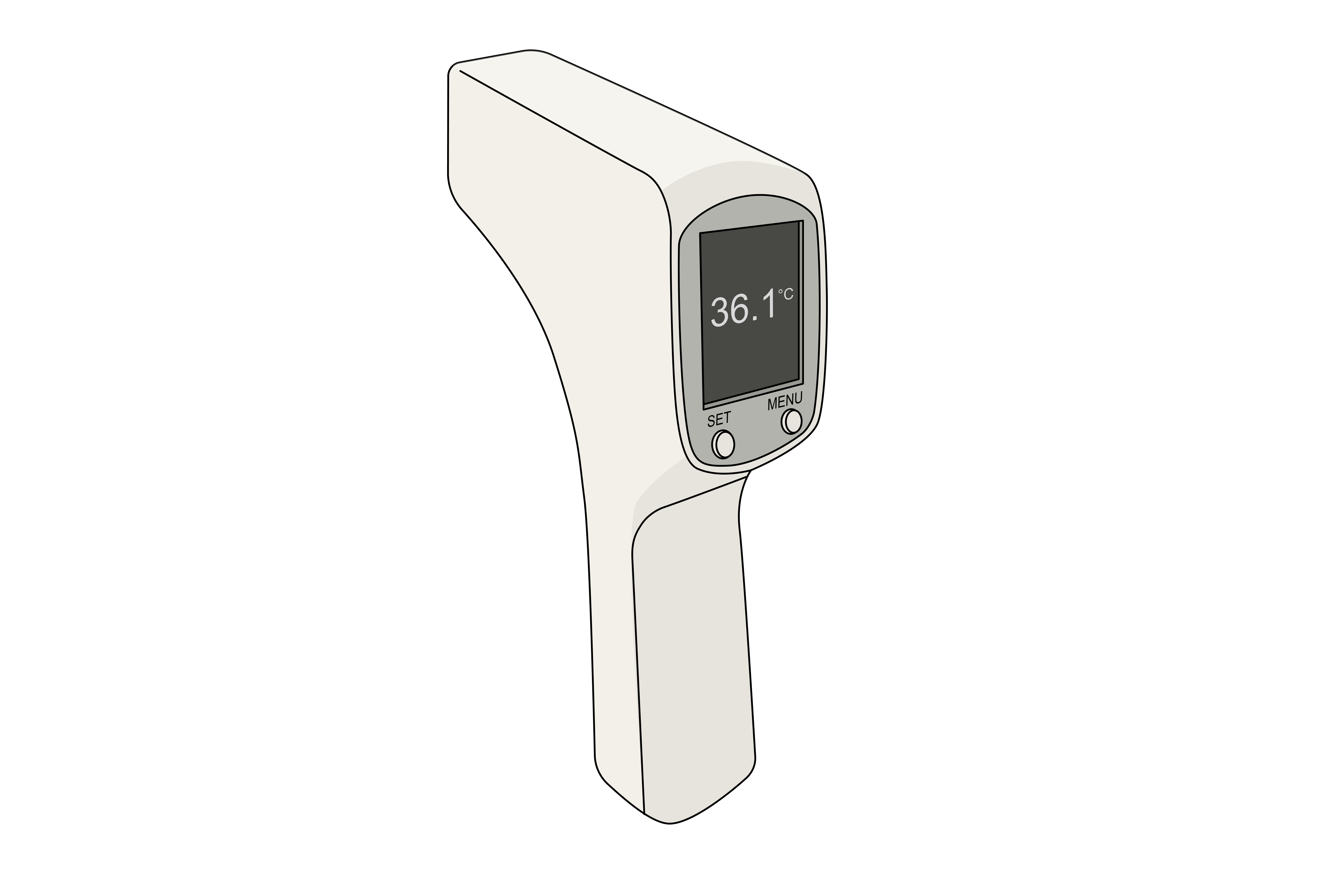
Infrared (IR), non-contact, clinical thermometer, digital, handheld, range 34 – 42 °C, including batteries.
Indicative Price 20.43 USD
GENERAL DESCRIPTION
Non-contact infrared clinical thermometer including batteries.
INTENDED USE
A hand-held, electrically-powered instrument designed to estimate the temperature of a site on the skin (e.g., axilla, forehead) by measurement of body infrared emissions at this particular point. It provides a method to determine temperature patterns or variations on the surface of the skin (e.g., due to differences in perfusion). This device may be used in the home. This is a reusable device.
TECHNICAL SPECIFICATIONS
Measurement of skin temperature trough infrared radiation detection
IR spectral response 6,000 – 14,000 nm.
Preferred measuring distance 3 - 7 cm.
Accuracy 0.2°C in the range of 35 - 42°C.
Graduation 0.1°C.
Out of range indication required.
Display visualisation of measured temperature, low battery, and malfunction.
Display easy to read in all levels of ambient light.
Digital read-out in Celsius or Fahrenheit, user selectable.
Low and high temperature indication.
Temperature measurement range 32 – 43 °C.
Response (measurement) time within 2 s.
Ready-to-use after switch-on within 10 s.
Automatic switch-off when not in use.
Automatic self-test on switch-on.
Visual and/or audible alert on switch-on, ready-to-use and measurement completed.
Full batteries allow for a minimum of 1,000 measurements.
The unit is light weight and designed to facilitate easy operation for prolonged durations.
Designed to withstand frequent cleaning and disinfection with hospital-grade products.
SUPPLIED WITH
Instructions for assembly, use and maintenance in English, French and Spanish.
1 x plastic protective case with hard outer shell.
1 x set of 2 suitable alkaline batteries.
SHELF LIFE
Not applicable.
ESTIMATED LIFE SPAN
Three to five years depending on the model supplied.
WARRANTY
Two years.
ENVIRONMENTAL CONDITIONS
Storage conditions: -20 - 60°C / 85% RH.
Operating conditions: 10 - 40°C / 30% - 85% RH.
Ingress Protection: IP21
Atmospheric pressure: 70kPa – 106kPa
WEIGHT AND VOLUME (Packaged)
Weight: 270 gr.
Volume: 1.23 dm³.
INSTALLATION / COMMISSIONING REQUIREMENTS
Other than installing the battery there are no installation or commissioning requirements.
TRAINING REQUIREMENTS
No user training will be required.
MAINTENANCE/USER REQUIREMENTS
Other than cleaning in accordance with manufactures instructions there are not specific maintenance requirements.
ALTERNATIVE PRODUCTS
S0481056, Thermometer, clinical, digital, no Li
COMPONENT OF A KIT
No.
QUALITY MANAGEMENT SYSTEM
- Manufacturer is certified for ISO 13485 Medical devices - Quality management systems - Requirements for regulatory purposes.
- Supplier is certified for ISO 9001 Quality management systems – Requirements.
MARKET CLEARANCE AND DEVICE CLASSIFICATION
CE certified under the EU MDD 93/42/EEC as Class IIa device.
SAFETY AND PRODUCT STANDARDS
- ISO 80601-2-56: 2017: Medical electrical equipment - Part 2-56: Particular requirements for basic safety and essential performance of clinical thermometers for body temperature.
- IEC 60601-1:2005 + A1:2012(E) Medical electrical equipment - Part 1: General requirements for basic safety and essential performance
- IEC 60601-1-2:2014 Medical electrical equipment - Part 1-2: General requirements for basic safety and essential performance - Collateral standard: Electromagnetic compatibility - Requirements and tests
- IEC 60601-1-6:2010 Medical electrical equipment - Part 1-6: General requirements for basic safety and essential performance - Collateral standard: Usability
- EN ISO 15223-1 (EN 980) Medical devices – Symbols to be used with medical device labels, labelling and information to be supplied – Part 1: General requirements.
NOMENCLATURE
GMDN Code: 17888
Related Products